Tetracaine & Lidocaine formulation
With the MTC-Y™ Carrier System
the treatment is controlled and sustained
Study design
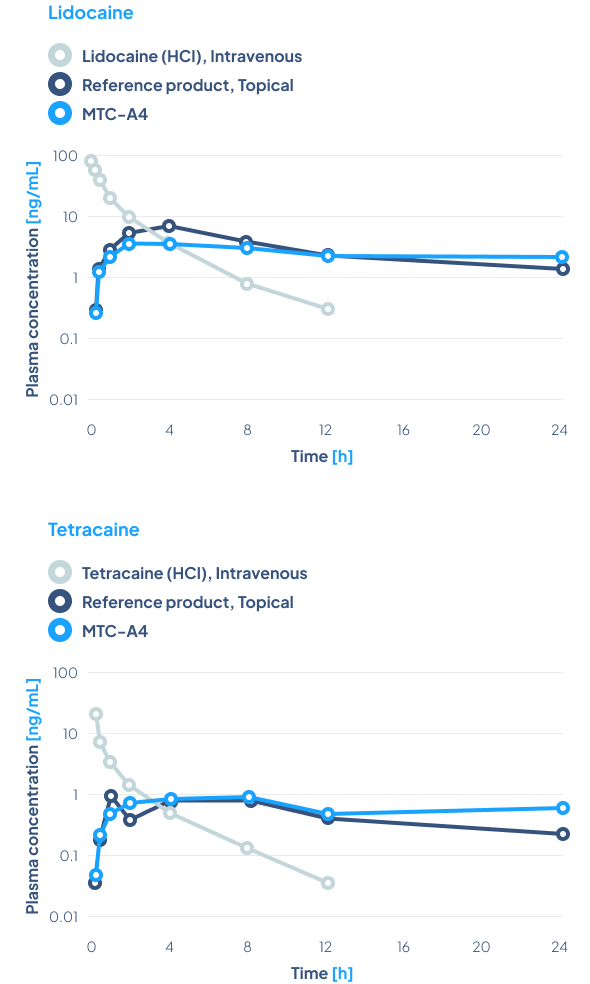
The concentration of the active substances, lidocaine and tetracaine, in the MTC-A4 product is 4% each, while in the reference product – Pliaglis – it is 7% each.
Results
The results show that using lower concentrations of active substances similar bioavailability of the same API was obtained. Furthermore, much more stable blood concentrations of the active substances released from MTC-A4 were obtained over a period of 24 hours, compared to the reference product.
COLLABORATIONS
Development collaborations
The MTC-Y™ carrier technology is the foundation of Biotts’ transdermal drug product development pipeline, but it’s also available to the pharmaceutical industry through contract development collaborations. Contact us or further information.