Safety of the MTC-Y Carrier
clinically confirmed
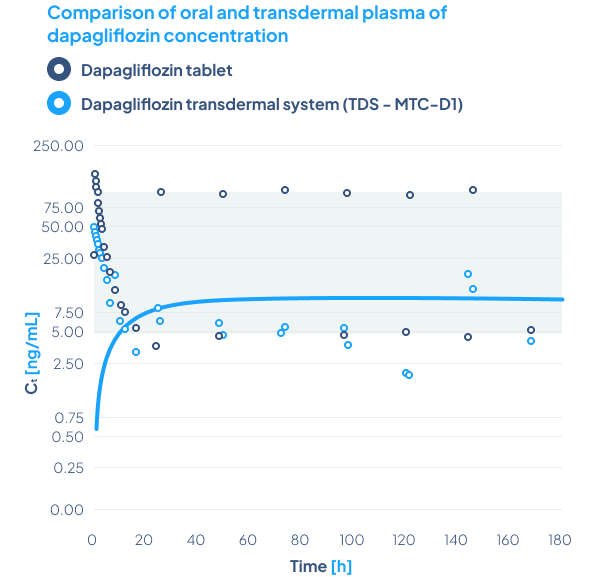
Comparison of oral and transdermal plasma of dapagliflozin concentration
Aim of the study
The primary objective of the phase 1 clinical study was to evaluate the safety, relative bioavailability and pharmacodynamic effect of a newly developed Dapagliflozin Transdermal System TTS MTC-D, after administration in healthy volunteers.
Results
Biotts’ transdermal TTS MTC-D system successfully delivered dapagliflozin through the skin and into the bloodstream.
The drug was well-tolerated, with mild side effects. The study confirmed the effectiveness of the MTC-Y carrier in delivering dapagliflozin, highlighting the potential of Biotts’ technology for diabetes treatment.
Scientific publications
Assessment of basic pharmacokinetic parameters of dapagliflozin in TTS formulations in male minipigs
Development collaborations
The MTC-Y™ carrier technology is the foundation of Biotts’ transdermal drug product development pipeline, but it’s also available to the pharmaceutical industry through contract development collaborations. Contact us or further information.