MTC-Y™
Semi-Solid Carrier
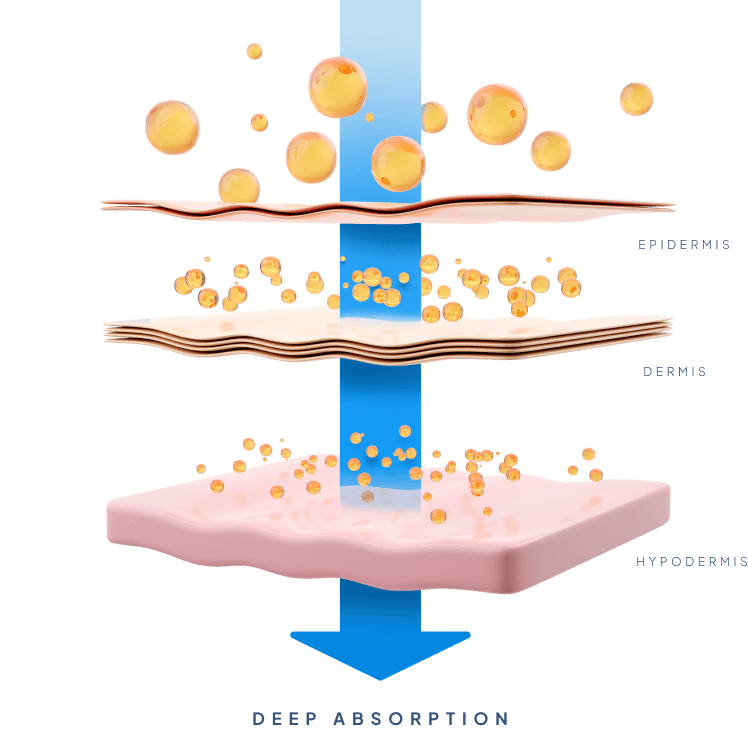
To be transdermal
Transdermal Drug Delivery has traditionally been limited by the skin’s tough and lipid rich outer layer known as the stratum corneum thus rendering the skin impermeable to most biopharmaceuticals; of the thousands of drugs currently approved by the FDA less than 30 are approved for transdermal delivery. So far, successful transdermal drug delivery has only been accomplished with small molecules (<500 Da.) that are moderately lipophilic. The development of Biotts’ proprietary MTC-Y™ technology has effectively removed these limitations, without the use of complicated devices or invasive technologies.
Biotts’ MTC-Y™ technology completely changes what is possible with Transdermal Drug Delivery, making it a much more feasible alternative. It offers the fast onset of action of an injectable, but at more constant and sustained drug levels and in a way that is much less invasive. Furthermore, it allows for reduced administration frequency and increased self-administration, and with that improved quality of life for patients. It also offers the convenience of an oral pill, but with a much more controlled and sustained release, and dramatically reduced side-effects and improved bioavailability.
Our technology
The mixture of excipients constituting the carrier of MTC-Y™ consists of components that ensure a simultaneous introduction of several active substances of different character and physicochemical properties into the body. Each of them can be introduced simultaneously in different polymorphic varieties, allowing for tailoring of the desired activity profile. It also enables significantly larger molecules – both lipophilic and hydrophilic – to penetrate through the skin barriers.
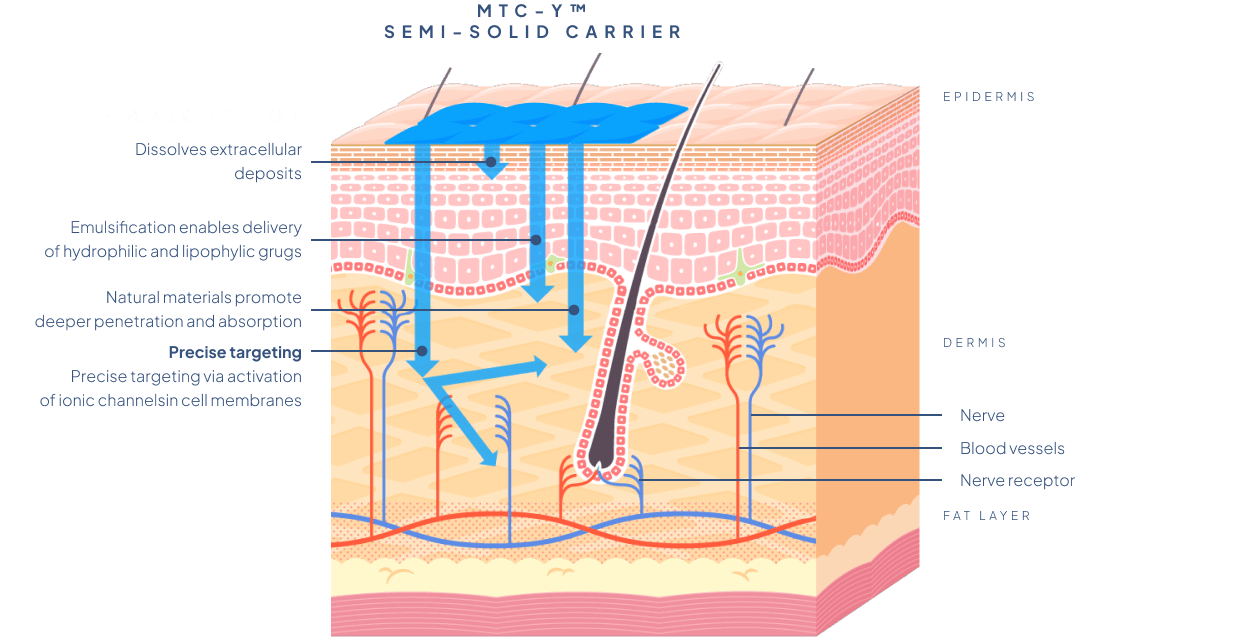
The MTC-Y™ Biotts system enables the transportation of one or more active substances, making it possilbe to design multi-component transdermal drugs. The MTC-Y™ carrier system is biocompatible with the human skin and non-toxic; it allows active ingredients to quickly penetrate the skin without irritating it.

particle [for now]
So far Biotts has generated Proof of Concept with a variety of molecules up to 6,000 Da. including several GLP-1 peptides like Liraglutide and Semaglutide, but there is no reason why we couldn’t go further. The possibilities of the MTC-Y™ technology seem endless.